You are here: Urology Textbook > Surgery (procedures) > Open nephroureterectomy
Open Nephroureterectomy: Step-by-Step Technique and Complications
Indications for Nephroureterectomy
Several different surgical approaches exist for nephroureterectomy, which have not been prospectively tested against each other. In retrospective comparisons, (robotic-assisted) laparoscopy lowers the complication rate and shortens the hospital stay (Hanna et al., 2012). All techniques have in common the surgical removal of the kidney, including the perirenal fat, the ureter with bladder cuff, and the associated lymph nodes (paracaval/paraaortal/pelvic, depending on tumor location). Consider open nephroureterectomy for advanced tumors (T3–4, N+) and large high-grade tumors (Simone et al., 2009).
Contraindications
Coagulation disorders. Further contraindications depend on the surgical risk due to the comorbidity of the patient, the renal function of the contralateral kidney, and the surgical procedure's impact on the patient's life expectancy. Kidney-sparing surgery is the first-choice for low-risk tumors if technically feasible. Kidney-sparing surgery must be considered even for high-risk tumors, if the preservation of kidney function is mandatory for the patient.
Preoperative patient preparation:
Perioperative antibiotic prophylaxis, perioperative Foley catheter with options for sterile bladder filling, gastric tube, consider epidural anesthesia for open surgery.
Extraperitoneal Pararectal Nephroureterectomy
Surgical approach:
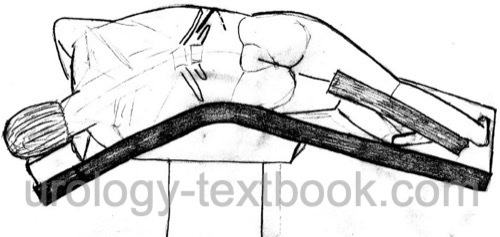
Flank incision from the 11th rib to the suprapubic region in a modified flank position.
Nephrectomy:
The tumor-bearing kidney is removed using an extraperitoneal technique; see section radical nephrectomy with a flank incision. The ureter is ligated early but not transected to avoid tumor cell seeding to the bladder. Aortic, interaortocaval, or caval lymphadenectomy is possible for proximal and mid-ureteral carcinoma.
Bladder cuff:
The ureter is mobilized with its surrounding fatty tissue to the urinary bladder. Ligation and transection of the medial umbilical ligament is helpful before the circular incision of the detrusor muscle to dissect the intramural ureter. Place stay sutures to the bladder wall before excising the ureter with the bladder cuff. A combined extravesical and intravesical dissection (using an anterior cystotomy) is also possible. Iliac and pelvic lymph nodes are removed for distal ureteral carcinoma.
Transurethral techniques to develop the bladder cuff have been published. However, the irrigation flow with extravasation increases the risk of tumor cell dissemination, and incomplete ureter excision is of concern. Transurethral techniques to minimize the surgical approach are not recommended.
Advantages:
Extraperitoneal procedure. Nephroureterectomy is possible without the transection of the ureter, and dissection of the distal ureter is oncologically safe.
Disadvantages:
Long surgical approach through the abdominal wall muscles.
Transperitoneal Nephroureterectomy
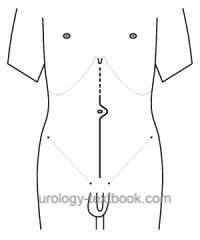 |
Surgical approach:
Midline incision from the xiphoid to below the umbilicus. Using a transperitoneal approach, the tumor-bearing kidney is removed together with the perirenal fat and regional lymph nodes, see section radical nephrectomy.
Advantages:
Nephroureterectomy is possible without transection of the ureter, and the dissection of the distal ureter is oncologically safe. The approach avoids transection of the abdominal wall muscles over a long distance. Midline laparotomy is the ideal approach for extensive lymphadenectomy.
Disadvantages:
Transperitoneal approach.
Nephroureterectomy With Two Incisions
Nephrectomy is done using a flank incision between the 11th and 12th rib, see section flank incision and extraperitoneal technique described above. The ureter is ligated and transected at the level of the iliac vessels.
 |
Distal ureteral resection and bladder cuff: after the wound closure of the flank incision, the patient is repositioned in supine. The distal ureter and bladder cuff are resected using a second incision (e.g., Pfannenstiel incision, extraperitoneal lower midline incision, or Gibson incision).
Advantages:
Extraperitoneal approach and less transection of the abdominal wall muscles due to two incisions.
Disadvantages:
It is unsuitable for the mid-third ureteral carcinoma, as the ureter has to be divided. The patient must be repositioned. Interaortocaval lymphadenectomy is more complicated.
Postoperative Care
Drains:
Drainage of the pelvic cavity for 1--2 days.
General Measures:
Early mobilization, intensive respiratory therapy, thrombosis prophylaxis, laboratory tests (hemoglobin, creatinine level), regular physical examination of the abdomen and incision wound.
Analgesics:
With a combination of NSAIDs and opioids. An epidural anesthesia facilitates postoperative pain management after open surgery.
Diet advancement:
Remove the nasogastric tube after surgery. Allow small sips of clear liquids after surgery. Increase amounts of clear liquids and allow yogurt or pudding on postoperative day 1 and 2. If the patients feels well, allow small amounts of solid food (appetite driven) starting postoperative day 3.
Drains and Catheters:
Wound drainage for 1–2 days. Removal of the bladder catheter after 5–7 days (with cystography in difficult cases).
Postoperative intravesical chemotherapy:
Tumor cell seeding during nephroureterectomy can result in urothelial carcinoma recurrence in the bladder. In the randomized ODMIT-C trial, a single postoperative intravesical chemotherapy with mitomycin C before catheter removal reduced the risk by 11% (16% vs. 27%). The number needed to treat is nine (OBrien et al., 2011).
Complications of Nephroureterectomy
See also complications of radical nephrectomy. Additional complications are persistent extravasation or urinoma due to an insufficient bladder cuff closure.
Risk of significant hemorrhage:
The risk of significant blood loss during radical nephrectomy is below 5%.
Injury of neighboring organs:
Liver injury, spleen injury (risk of splenectomy), paralytic ileus, bowel injury, peritonitis, pancreatic tail injury with fistula, pneumothorax, and chylous fistula due to injury of intestinal lymphatic vessels.
General complications:
Wound infection, heart attack, stroke, heart failure, thrombosis, pulmonary embolism, atelectasis, pneumonia, acute renal failure.
Mortality:
Mortality due to bleeding, cardiovascular diseases, arrhythmia, acute renal failure, and pulmonary embolism is around 1–2%, with advanced tumor stage being the leading risk factor.
| Ureteroscopy | Index | Laparoscopic nephroureterectomy |
Index: 1–9 A B C D E F G H I J K L M N O P Q R S T U V W X Y Z
References
Hanna, N.; Sun, M.; Trinh, Q.; Hansen, J.; Bianchi, M.;
Montorsi, F.; Shariat, S. F.; Graefen, M.; Perrotte, P. & Karakiewicz, P.
I.
Propensity-score-matched comparison of perioperative outcomes
between open and laparoscopic nephroureterectomy: a national series.
Eur
Urol, 2012, 61, 715-721.
O'Brien, T.; Ray, E.; Singh, R.; Coker, B.; Beard, R. &
of Urological Surgeons Section of Oncology, B. A.
Prevention of bladder
tumours after nephroureterectomy for primary upper urinary tract
urothelial carcinoma: a prospective, multicentre, randomised clinical
trial of a single postoperative intravesical dose of mitomycin C (the
ODMIT-C Trial).
Eur Urol, 2011, 60, 703-710.
G. Simone et al., “Laparoscopic versus open nephroureterectomy: perioperative and oncologic outcomes from a randomised prospective study.,” Eur Urol, vol. 56, no. 3, pp. 520–526, 2009, doi: 10.1016/j.eururo.2009.06.013.
 Deutsche Version: offen-chirurgische und laparoskopische Nephroureterektomie
Deutsche Version: offen-chirurgische und laparoskopische Nephroureterektomie
Urology-Textbook.com – Choose the Ad-Free, Professional Resource
This website is designed for physicians and medical professionals. It presents diseases of the genital organs through detailed text and images. Some content may not be suitable for children or sensitive readers. Many illustrations are available exclusively to Steady members. Are you a physician and interested in supporting this project? Join Steady to unlock full access to all images and enjoy an ad-free experience. Try it free for 7 days—no obligation.
New release: The first edition of the Urology Textbook as an e-book—ideal for offline reading and quick reference. With over 1300 pages and hundreds of illustrations, it’s the perfect companion for residents and medical students. After your 7-day trial has ended, you will receive a download link for your exclusive e-book.