You are here: Urology Textbook > Drugs in Urology > Mirabegron
Mirabegron: Mechanism of Action, Adverse Effects, Contraindications, and Dosing
Mechanism of Action of Mirabegron
Mirabegron is a β3-adrenergic receptor agonist. In the bladder, mirabegron increases intracellular cAMP and relaxes the detrusor smooth muscle, which increases the functional bladder capacity.
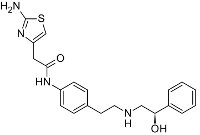
Urologic Indications for Mirabegron:
- Symptomatic treatment of urgency symptoms associated with overactive bladder (OAB) in adults. In pivotal registration trials, mirabegron reduced the number of incontinence episodes and micturitions and increased mean voided volume (compared with placebo and tolterodine) (Khullar et al., 2013) (Nitti et al., 2013) (Herschorn et al., 2013).
- Neurogenic detrusor overactivity in children older than 3 years.
- Off-label use: neurogenic detrusor overactivity in adults.
Pharmacokinetics of Mirabegron
The oral bioavailability is approximately 35%, and patients may take the medication with or without food. The liver metabolizes mirabegron via multiple pathways, and the inactive and some active metabolites are eliminated with the urine and feces. The terminal half-life is about 50 hours, and patients typically reach steady state after 7 days.
Adverse Effects of Mirabegron
Mirabegron can increase blood pressure, it is essential to measure blood pressure before treatment and monitor it periodically during therapy. Long-term cardiovascular safety has been demonstrated in several studies; however, caution is warrented in patients with significant cardiovascular risks.
- Common (1–10%): tachycardia (1%), urinary tract infections, headache or dizziness, nausea, constipation, or diarrhea.
- Uncommon to rare: blood pressure elevation up to hypertensive crisis; palpitations or arrhythmias (rarely atrial fibrillation); angioedema or allergic reactions; dyspepsia or gastritis; elevated liver enzymes; urinary retention (especially in patients with clinically relevant bladder outlet obstruction or when combined with antimuscarinics).
Drug Interactions:
Strong CYP3A inhibitors such as itraconazole, ketoconazole, ritonavir, or clarithromycin can increase mirabegron levels. When patients take strong CYP3A inhibitors, limit the dose to 25 mg once daily in patients with mild hepatic impairment or mild-to-moderate renal impairment. Do not combine mirabegron with CYP3A inhibitors in patients with severe renal or hepatic impairment.
Mirabegron is a moderate CYP2D6 inhibitor and a P-glycoprotein (P-gp) inhibitor, so plasma concentrations of relevant substrates can increase. Monitor the substrates with a narrow therapeutic index and consider dose adjustments (e.g., digoxin).
Combination therapy with antimuscarinics, for example, with solifenacin (Abrams et al., 2015), can provide greater symptom improvement than monotherapy; however, it can increase constipation and dry mouth and may increase the risk of urinary retention, particularly in patients with bladder outlet obstruction.
Contraindications of Mirabegron
Severe uncontrolled hypertension (systolic ≥180 mmHg or diastolic ≥110 mmHg). Severe hepatic impairment (Child-Pugh C). End-stage renal disease (GFR <15 ml/min). Hypersensitivity to the active ingredient. Pregnancy and breastfeeding.
Dosing of Mirabegron
- Adults: 50 mg once daily. Reduce the dose to 25 mg by mouth in severe renal impairment (GFR 15–29 ml/min) or moderate hepatic impairment (Child-Pugh B).
- Children: Dose mirabegron by body weight using an extended-release suspension or tablets for higher body weights.
Brand Names:
Betmiga in Europe, Myrbetriq in the USA.
| Anticholinergics | Index | Vibegron |
Index: 1–9 A B C D E F G H I J K L M N O P Q R S T U V W X Y Z
References
Herschorn, S. u.a.
A phase III, randomized, double-blind,
parallel-group, placebo-controlled, multicentre study to assess the
efficacy and safety of the β₃ adrenoceptor agonist, mirabegron, in
patients with symptoms of overactive bladder.
Urology 2013, 82,
313-320.
Khullar, V.; Amarenco, G.; Angulo, J. C.; Cambronero,
J.; Høye, K.; Milsom, I.; Radziszewski, P.; Rechberger, T.; Boerrigter,
P.; Drogendijk, T.; Wooning, M. & Chapple, C.
Efficacy and
tolerability of mirabegron, a β(3)-adrenoceptor agonist, in patients with
overactive bladder: results from a randomised European-Australian phase 3
trial.
Eur Urol 2013, 63, 283-295.
Nitti, V. W.; Auerbach, S.; Martin, N.; Calhoun, A.; Lee, M. & Herschorn, S.
Results
of a randomized phase III trial of mirabegron in patients with overactive
bladder.
J Urol 2013, 189, 1388-1395.
 Deutsche Version: Mechanismus, Nebenwirkungen und Kontraindikationen von Mirabegron
Deutsche Version: Mechanismus, Nebenwirkungen und Kontraindikationen von Mirabegron
Urology-Textbook.com – Choose the Ad-Free, Professional Resource
This website is designed for physicians and medical professionals. It presents diseases of the genital organs through detailed text and images. Some content may not be suitable for children or sensitive readers. Many illustrations are available exclusively to Steady members. Are you a physician and interested in supporting this project? Join Steady to unlock full access to all images and enjoy an ad-free experience. Try it free for 7 days—no obligation.
New release: The first edition of the Urology Textbook as an e-book—ideal for offline reading and quick reference. With over 1300 pages and hundreds of illustrations, it’s the perfect companion for residents and medical students. After your 7-day trial has ended, you will receive a download link for your exclusive e-book.